| Element | Part | Count | Atomic Mass | Contribution |
|---|
Best Theoretical Percentage of Water Calculator for Hydrates (2026 Step-by-Step Guide)
Staring at a hydrate formula might leave you wondering where to even start. That moment when the numbers seem to blur together? Totally normal. Picture yourself flipping through pages, needing clarity fast. Maybe you are double-checking work, maybe just practicing. Either way, knowing exactly how much water hides inside those crystals matters. A tool that gives you the theoretical water percentage helps quiet the noise. Suddenly, what looked messy makes sense. It cuts through guesswork like a clear signal in static.
Picture this – what even is a hydrate? Turns out, it’s just a compound holding onto water molecules like they’re old friends. Figuring out how much of that weight comes from water isn’t just textbook stuff – it shows up where it counts. Step one: break down the molar masses, piece by piece.
Take the water part. Then the whole structure. Divide, multiply by 100 – you’ve got your percent. Sounds tidy when you see it done. That’s why actual number-crunching cases help. One with copper sulfate. Maybe another using magnesium something. A neat little ratio trick sticks better than rules. Tools? Yeah, some online calculators skip the grunt work. Not magic – just math on autopilot.
What Is a Hydrate in Chemistry?
Water sticks inside some crystals – this trapped moisture has a name. Think of it like tiny droplets caught in a cage made by the solid stuff around them. That whole mix – the crystal plus the H₂O tucked away – is what scientists call a hydrate. Those hidden water bits aren’t just sitting there; they’re part of the shape, giving each lump its unique form.
A well-known case is copper(II) sulfate pentahydrate, shown as CuSO₄·5H₂O. The ·5H₂O bit means five water molecules stick to each copper sulfate unit. Because that’s how it forms.
Truth is, hydrates seem parched at first glance. Yet deep inside, they hold onto plenty of water molecules. This hidden moisture makes calculating their exact water content crucial work in lab analysis.
Among everyday hydrates you might come across are these
Copper(II) sulfate pentahydrate is CuSO₄·5H₂O
MgSO₄·7H₂O – Magnesium sulfate heptahydrate (Epsom salt)
Na₂CO₃·10H₂O – Sodium carbonate decahydrate (washing soda)
CaCl₂·2H₂O – Calcium chloride dihydrate
Figuring out how much of these substances is just water lets researchers check quality, work out output amounts, yet shape studies the right way.
Why Use a Theoretical Percentage of Water Calculator for Hydrates?
Truth is, working out the numbers yourself isn’t impossible. Still, what’s the point if a tool built for finding water percentages in hydrates finishes in moments?
This is the reason such math holds weight: it shapes choices without drawing attention
Start by heating the hydrate so water escapes into the air. Its weight drops once moisture leaves the sample behind. That change gets measured with care afterward. A calculation predicts how much should vanish if everything were ideal. The real result lines up close when purity holds true. Matching numbers mean no extras messed up the composition.
Figuring out how much water is in a compound often shows up on chemistry tests. Sometimes you get a chemical formula, then need to work through the math behind it.
Water levels matter in factories and drug production because they shape how long items stay usable. Stability of goods often ties directly to exact moisture measurements during making things. Length of storage hinges on controlling wetness right from the start.
Most mistakes vanish when steps come straight from a trusted method. A steady approach beats guessing each time. Precision shows up easiest when numbers follow a set path. Following rules closely keeps results honest. Tools like calculators guard against slips. Clear structure handles what memory cannot. Complexity fades once routine takes over.
The Core Formula: How to Calculate Theoretical % Water in a Hydrate
Here’s the formula you need to know:
% Water = (Mass of Water in Formula / Molar Mass of Hydrate) × 100
Let’s break that down into plain English:
- Find the molar mass of the water portion (number of H₂O molecules × 18.015 g/mol)
- Find the total molar mass of the entire hydrate compound
- Divide the water mass by the total mass
- Multiply by 100 to get a percentage
It sounds simple, right? It is — once you practice it a few times.
Step-by-Step Guide to Using the Theoretical Percentage of Water Calculator for Hydrates
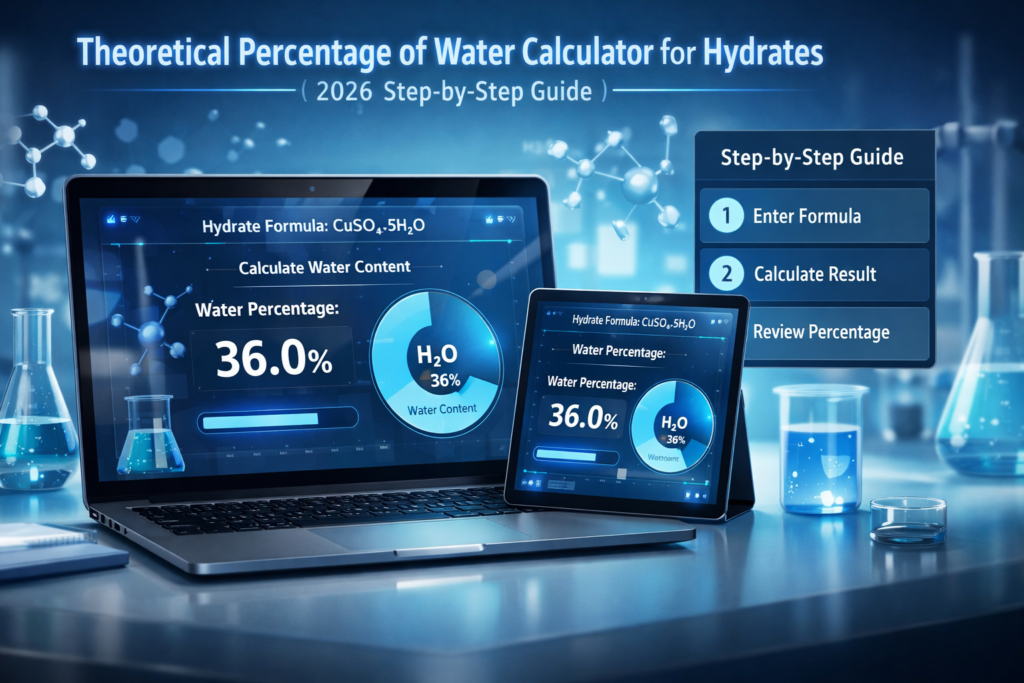
Let’s walk through a full example using CuSO₄·5H₂O (copper sulfate pentahydrate).
Step 1: Identify the Hydrate Formula
Your formula is: CuSO₄·5H₂O
This means:
- 1 Cu (Copper)
- 1 S (Sulfur)
- 4 O (Oxygen) — from the sulfate
- 5 H₂O — water molecules
Step 2: Calculate the Molar Mass of the Anhydrous Salt
Find the molar mass of CuSO₄:
| Element | Atoms | Atomic Mass | Total |
|---|---|---|---|
| Cu | 1 | 63.55 g/mol | 63.55 |
| S | 1 | 32.07 g/mol | 32.07 |
| O | 4 | 16.00 g/mol | 64.00 |
| CuSO₄ Total | — | — | 159.62 g/mol |
Step 3: Calculate the Molar Mass of the Water Portion
5 H₂O = 5 × 18.015 = 90.075 g/mol
Step 4: Calculate the Total Molar Mass of the Hydrate
Total = 159.62 + 90.075 = 249.695 g/mol
Step 5: Apply the Formula
% Water = (90.075 / 249.695) × 100 = 36.08%
So, CuSO₄·5H₂O is 36.08% water by mass. That’s more than a third of its total weight — just from water molecules!
How the Theoretical Percentage of Water Calculator for Hydrates Works Online
Several online chemistry tools let you enter a hydrate formula and instantly get the theoretical water percentage. Here’s what a good calculator typically asks for:
- Chemical formula of the hydrate (e.g., CuSO₄·5H₂O)
- Number of water molecules (n in the formula)
It then automatically:
- Parses the formula elements
- Pulls atomic masses from a built-in database
- Calculates total molar mass
- Outputs the theoretical % water
What I’ve found is that most students prefer online calculators for quick homework checks, while understanding the manual method is essential for exams and lab reports.
Common Mistakes When Calculating % Water in Hydrates
Let’s be real — even experienced chemistry students make these errors. Watch out for:
- Using the wrong atomic mass — Always use the standard atomic weight (e.g., H = 1.008, not 1)
- Forgetting to multiply water molecules — If it’s 5H₂O, the mass is 5 × 18.015, not just 18.015
- Confusing anhydrous vs. hydrate mass — The denominator must be the full hydrate molar mass
- Rounding too early — Keep extra decimal places until the final answer
One more thing: make sure you’re reading the hydrate formula correctly. The dot (·) in CuSO₄·5H₂O separates the salt from the water — it’s not a multiplication sign in the traditional sense.
Practice Examples: Test Your Skills
Try these on your own before checking the answers:
Example 1: BaCl₂·2H₂O (Barium chloride dihydrate)
- Molar mass of BaCl₂ = 208.23 g/mol
- Water: 2 × 18.015 = 36.03 g/mol
- Total = 244.26 g/mol
- % Water = (36.03 / 244.26) × 100 = 14.75%
Example 2: Na₂SO₄·10H₂O (Sodium sulfate decahydrate)
- Molar mass of Na₂SO₄ = 142.04 g/mol
- Water: 10 × 18.015 = 180.15 g/mol
- Total = 322.19 g/mol
- % Water = (180.15 / 322.19) × 100 = 55.92%
That second one is wild, isn’t it? Over half of sodium sulfate decahydrate is just water.
Comparing Theoretical vs. Experimental % Water
In a real chemistry lab, you’ll heat a hydrate sample to remove the water, weigh before and after, and calculate the experimental percentage of water. Then you compare it to the theoretical value.
| Parameter | Theoretical % Water | Experimental % Water |
|---|---|---|
| Based on | Chemical formula | Actual lab measurement |
| Always exact? | Yes | Varies (human/equipment error) |
| Used for | Identifying compounds | Verifying purity |
| Calculation method | Formula + molar mass | (Mass lost / Original mass) × 100 |
A small difference between the two is normal. A large difference? That usually means your sample wasn’t pure, or the heating wasn’t complete.
Tips for Getting Accurate Results
Whether you’re using a theoretical percentage of water calculator for hydrates or doing the math manually, here are some practical tips:
- Double-check your formula — Even one wrong digit changes everything
- Use updated atomic masses — The IUPAC publishes regular updates
- For lab work: Heat the hydrate slowly to avoid spattering
- Round only at the end — Keep at least 4 significant figures throughout
- Cross-check with a secondary calculator — Always verify important results
FAQ Section
Q1: What is the theoretical percentage of water in a hydrate? It is the calculated percentage of the hydrate’s total molar mass that comes from water molecules, based purely on its chemical formula — not from experimental measurement.
Q2: How do I use a theoretical percentage of water calculator for hydrates? Enter the hydrate’s chemical formula (e.g., CuSO₄·5H₂O) into the calculator. It will automatically compute the molar mass of both the water portion and the full compound, then output the theoretical water percentage.
Q3: What formula is used to calculate % water in a hydrate? The formula is: % Water = (Molar Mass of Water in Formula ÷ Molar Mass of Hydrate) × 100
Q4: Why is the theoretical percentage of water different from the experimental value? The theoretical value is based on a perfect chemical formula. The experimental value comes from real lab measurements, which can vary due to incomplete heating, impurities in the sample, or measurement errors.
Q5: What is the molar mass of water used in hydrate calculations? The molar mass of water (H₂O) is 18.015 g/mol — calculated from 2 × 1.008 (hydrogen) + 15.999 (oxygen).
Q6: Can I use the same method for all hydrates? Yes! The formula works for any hydrate as long as you have the correct chemical formula and accurate atomic masses for all elements involved.
